Unfortunately, we should expect most PBMs and plan sponsors to embrace the high-list/high-rebate version. The plans that adopt the higher-priced biosimilar will get bigger rebates, while patients with coinsurance and deductibles end up paying more out-of-pocket.
As Mark Twain said: “History doesn’t repeat itself—but it often rhymes.” So, below, I revisit the surprising market dynamics for the interchangeable insulin biosimilars and highlight Amgen’s experience lowering the list price of its PCSK9i product. The story is more complex than you might expect, so perhaps there is hope that low-list-price versions will connect.
AMGEN AGAIN
Amgen is major biosimilar player, so it’s fitting that the company launched the first Humira biosimilar. Here’s the official press release: Amjevita™ (adalimumab-atto), First Biosimilar To Humira®, Now Available In The United States. As the release notes, Amjevita will be available at different two wholesale acquisition cost (WAC) list prices: a price that is 5% below Humira’s current WAC, and a price that is 55% below Humira’s current WAC.
On its recent earnings call, Amgen was unambiguous about its rationale for the dual-pricing strategy:
“With respect to the two list price approach that we've employed here at this launch, this is really to address the complexity of the U.S. market. Pharmacy benefit managers have a business model that requires that they negotiate rebates with manufacturers, and so they would prefer a high list price and negotiate rebates to net the price down and then pass those rebates through to their upstream employer clients.”While true, market access conspiracy theorists will also see Amgen signaling the minimum rebate level for its Humira biosimilars. I presume the later entrants will get the message.
Also notable: Amgen plans to provide patient hub services for its biosimilars that are comparable to a brand-name drug. From the earnings call:
“In addition, of course, we provide nurse support for patients. And then while it wasn't asked, I think it's also important to note that we are providing financial assistance, support and reimbursement support for both prescribers and patients as we launch the product. So really a full suite of services and support that you would expect for a branded launch being applied to the launch of the first biosimilar, adalimumab, to launch in the U.S., that is AMJEVITA.”FYI, Amgen has stated that the readout from Amjevita’s Phase III interchangeability study will be available in the first half of 2023.
HIGH-PRICE SEMGLEE FOR THEE
Let’s revisit the three largest PBMs’ formulary strategies for the long-acting insulin market. For background, see:
- Why PBMs and Payers Are Embracing Insulin Biosimilars with Higher Prices—And What That Means for Humira (November 2021)
- The Big Three PBMs’ 2023 Formulary Exclusions: Observations on Insulin, Humira, and Biosimilars (January 2023)
- Semglee (insulin glargine-yfgn) injection, a branded interchangeable product with a high list price
- Insulin Glargine (insulin glargine-yfgn) injection, an unbranded, authorized interchangeable biosimilar with a low list price
- CVS Caremark prefers Basaglar, a non-interchangeable follow-on biologic, and blocks Lantus. Neither Semglee nor its unbranded version is listed on the Caremark formulary.
- Express Scripts prefers the high-list price Semglee and excludes both Lantus and the low-priced, unbranded interchangeable biosimilar. (The unbranded version is preferred on the PBM’s little-used Flex formulary.)
- OptumRx prefers the Lantus reference product and excludes Semglee and the unbranded biosimilar.
[Click to Enlarge]
Here’s what happened:
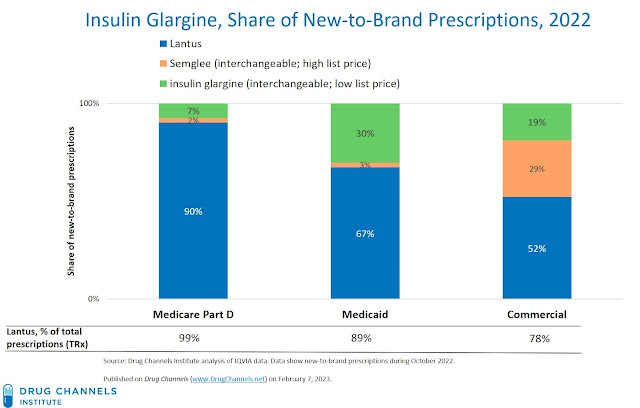
- Commercial payers adopted the biosimilars, but preferred the high-list/high-rebate biosimilar over its low-list price version. The interchangeable biosimilars gained 48% of new-to-brand prescriptions, of which 60% (29%/48%) were the high-list/high-rebate version.
These adoption rates are generally consistent with the PBMs’ commercial formulary exclusion decisions above. Given the large existing patient base, the reference Lantus product retained 78% of the total commercial market almost one year after the biosimilars launched. But it’s notable that at least a subset of plans were willing to adopt the low-WAC versions.
- Medicare Part D plans continued to preference the high-list/high-rebate reference Lantus product, which retained 99% of the total market.
- Medicaid plans were more likely to adopt the low-list biosimilar, but retained significant use of the reference products. Presumably, the net price of Lantus to Medicaid was low enough that the reference product remained the most attractive option.
However, I’m puzzled by Medicaid programs’ adoption of the high-list biosimilar. Perhaps some Medicare Advantage tomfoolery?
Note that the net, post-rebate price of Lantus has declined significantly. From 2012 through 2021, the net price of Lantus declined by 62%, even as its list price rose. I estimate that non-Medicaid rebates and discounts for Lantus are more than 80% off its list price. These declines were due to competition from non-interchangeable Basaglar as well as the interchangeable biosimilars.
Undoubtedly, some patients have been paying too much for their biosimilar prescriptions. The plans and PBMs who adopted the higher-priced version have been getting bigger rebates, while patients with coinsurance and deductibles have paid more out-of-pocket.
REPATHA MATHA
In case you have forgotten, Amgen has experience with alternate list price strategies.
Amgen’s Repatha (evolocumab), a PCSK9 inhibitor, was approved in 2015. In 2018, Amgen launched a second version of the product with a WAC list price that was more than 60% below the original, high-list price version. The lower price reflected the post-rebate net cost of this therapy. The high-list/high-rebate version was subsequently discontinued. At the time, I wrote: “This is a huge pro-patient move, especially for those whose coinsurance and deductibles expose them to the artificial, pre-rebate list price.”
Guess what happened? By 2020, a typical patient’s average per-prescription out-of-pocket obligation went from $157 for the high-list price version to $70 for the low-list price version. Patients with coinsurance experienced the largest reductions in out-of-pocket costs. Average pharmacy reimbursement fell, from $1,500 per prescription to $540. (Full details in this fascinating paper.)
UNENTICING PRICING?
The Humira biosimilar market will be much more complex than the insulin and PCSK9i markets. The forthcoming Humira biosimilars will have differences in formulation and concentration. Some—but likely not all—of the other Humira biosimilars will have interchangeability with Humira.
PBMs have been noticeably reluctant to reveal their formulary coverage decisions for Amjevita’s high vs. low priced products:
- When asked about Humira biosimilars during its Q4 earnings call last week, Cigna’s CEO claimed that Express Scripts will “preserve and expand choice with aligned economics back to our clients and for the benefit of our customers and patients.” This statement appears to be content-free. Nonetheless, we still don’t know whether Express Scripts will block the low-list-price Amjevita on its main formulary.
- OptumRx’s CEO declared (in a LinkedIn post, for some reason) that Humira biosimilars are “the biggest opportunities in years to help lower health care costs”—but then noted that the PBM’s clients “can include both Amjevita list price options on our commercial formulary.” Yikes.
Looks like 2023 could be even weirder than I expected. Let the games begin!


No comments:
Post a Comment