As we explain below, this accelerating adoption also boosts profits for pharmaceutical wholesalers and specialty distributors—particularly when these channel participants can influence which biosimilar version a provider utilizes.
Below, we share our estimates for wholesalers’ gross margins for brand-name, generic, and biosimilar drugs. We then explain the channel dynamics behind biosimilar and generic drugs’ profits. We estimate that for 2022, generics and biosimilars accounted for only about 15% of wholesalers’ U.S. drug distribution revenues, but 77% of total gross profits from U.S. drug distribution.
Read on for the fun financial facts.
Today’s article is adapted from our new 2022-23 Economic Report on Pharmaceutical Wholesalers and Specialty Distributors. Special discounted pricing ends this Friday (October 21, 2022).
MARGIN MATH
Gross profit equals the revenues that a wholesaler receives minus the cost of products (net of discounts and returns) bought from a manufacturer or (rarely) another wholesaler. Gross profit therefore measures the portion of revenues available for the operating expenses and operating profit of a wholesaler. It reflects how much a wholesaler is compensated for taking on specific tasks and functions in the distribution system. Gross margin expresses gross profit as a percentage of revenues.
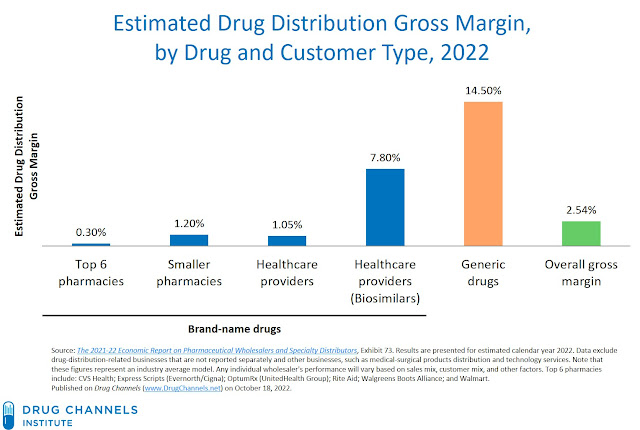
Consider the the Big Three public wholesalers—AmerisourceBergen, Cardinal Health, and McKesson. The chart below summarizes our estimates of these wholesalers’ 2022 average gross margins for brand-name and biosimilar drugs—sold to three customer types—and generic drugs.
[Click to Enlarge]
A few observations:
- The overall gross margin may seem low compared with other industries. However, the big three wholesalers' revenues will be more than $600 billion in 2022, so the low margin translates into almost $16 billion in gross profits from distirbution.
- The gross margin percentage for generic drugs is much higher than the margin for brand-name drugs. As we explain in Section 4.2.4. of our new 2022-23 wholesaler economic report, generic drugs’ higher margin derives from the channel’s enhanced bargaining position with generic manufacturers compared with brand-name manufacturers. A higher gross margin percentage also compensates a wholesaler for the lower base upon which the margin is computed. (There are also balance sheet effects, which are beyond the scope of today's article.)
- For brand-name drugs, overall gross margins are low, although these products have much higher prices. There are complex margin dynamics between larger vs. smaller customers, and larger vs. smaller manufacturers.
The biosimilar profit story is more complex. Per the chart above, we estimate that wholesalers’ biosimilar gross margin sits between the brand-name and generic figures.
Biosimilar products are alternative versions of an already-approved biological product, i.e., the single-source, brand-name reference drug. Twenty-eight of the 39 FDA-approved biosimilars are provider-administered products covered primarily under a patient’s medical benefit and sold by wholesalers to physician offices, clinics, and hospital outpatient locations.
These products are marketed as brand alternatives rather than as interchangeable generics. Each branded biosimilar therefore establishes its own position on the formularies of commercial third-party payers, which often favor one biosimilar over another.
Consequently, a wholesaler cannot credibly threaten to exclude a biosimilar, because that product may have coverage at one or more health plans. To provide a complete selection to physicians and providers, wholesalers are instead forced to stock the innovator biologic reference product along with every biosimilar version.
DISTRIBUTION DYNAMICS
The market and competitive dynamics of provider-administered biosimilars enable wholesalers’ gross margins from these products to be higher than brand-name drugs, but lower than generic drugs.
As with interchangeable generic drugs, wholesalers cannot increase the overall demand for a therapeutic class of drugs. However, wholesalers can use pricing and availability in an attempt to influence a provider’s selection of one manufacturer’s biosimilar drug over another manufacturer’s version.
The wholesalers’ influence is greatest when:
- There are multiple biosimilar products of a reference drug. A growing number of biological drugs now compete with multiple biosimilar versions, including Herceptin (5 versions), Neulasta (6), Remicade (3), and Rituxan (4). This competition has allowed wholesalers to extract additional discounts and rebates from biosimilar manufacturers.
- A payer has not mandated use of an innovator product or a specific biosimilar. Formulary designs have complicated wholesalers’ distribution operations and limit their negotiating leverage. The wholesalers cannot credibly threaten to exclude a biosimilar, because that product may have coverage at one or more health plans. Over the past few years, payers have begun to alter their strategies to favor biosimilars and remove restrictions on provider choices, thereby giving prescribers more flexibility in product selection. However, these programs vary significantly among payer and products.
- Providers do not have financial disincentives to use biosimilars. Buy-and-bill reimbursement dynamics impact the substitution of provider-administered biosimilars, although these effects are not straightforward. There are also evolving complexities in reimbursement issues for commercial vs. Medicare Part B, and for 340B hospitals vs. physician offices and non-340B entities. The Inflation Reduction Act of 2022 will add new twists with a temporary increase in the add-on payment rate for biosimilars and an expected reduction in overall add-on payments for government-negotiated drugs.
Wholesalers also try to leverage differences in list prices between reference products and biosimilars to negotiate higher fees from manufacturers. Wholesalers’ buy-side fees and payment discounts for biosimilars are still computed based on the product’s wholesale acquisition cost (WAC) list price. They therefore try to negotiate distribution fee rates that fully or partially offset the effect of biosimilar's lower list prices and maintain a consistent buy-side fee rate per unit. Otherwise, they have disincentives to stock biosimilars or supply them to their customers. (The ultimate dollar rate for the biosimilar negotiated between the manufacturer and wholesaler may or may not be equivalent to the rate paid for the reference product.)
OUTLOOK
Although wholesaler revenues are linked most closely to sales of brand-name drugs, the majority of wholesalers’ gross profits comes from generic and biosimilar drugs. The generics and biosimilar market continues to expand, so we expect that wholesalers’ influence—and compensation—will also grow.

No comments:
Post a Comment